Calcium iodate
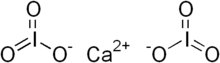 | |
| Names | |
|---|---|
| IUPAC name Calcium diiodate | |
| Other names Lautarite | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.029.265 |
| EC Number |
|
| E number | E916 (glazing agents, ...) |
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | Ca(IO3)2 |
| Molar mass | 389.88 g/mol (anhydrous) 407.90 g/mol (monohydrate) |
| Appearance | white solid |
| Density | 4.519 g/cm3 (monohydrate) |
| Melting point | 540 °C (1,004 °F; 813 K) (monohydrate) |
| Boiling point | decomposes |
Solubility in water | 0.09 g/100 mL (0 °C) 0.24 g/100 mL (20 °C) 0.67 g/100 mL (90 °C) |
Solubility product (Ksp) | 6.47×10−6 |
| Solubility | soluble in nitric acid insoluble in alcohol |
| -101.4·10−6 cm3/mol | |
| Structure | |
| monoclinic (anhydrous) cubic (monohydrate) orthorhombic (hexahydrate) | |
| Hazards | |
| Flash point | non-flammable |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).  N verify (what is N verify (what is  Y Y N ?) N ?) Infobox references | |
Calcium iodate is any of two inorganic compounds with the formula Ca(IO3)2(H2O)x, where x = 0 or 1. Both are colourless salts that occur as the minerals lautarite and bruggenite, respectively. A third mineral form of calcium iodate is dietzeite, a salt containing chromate with the formula Ca2(IO3)2CrO4.[1] These minerals are the most common compounds containing iodate.
Production and uses
Lautarite, described as the most important mineral source of iodine, is mined in the Atacama Desert.[1] Processing of the ore entails reduction of its aqueous extracts with sodium bisulfite to give sodium iodide. This comproportionation reaction is a major source of the sodium iodide.[1]
Calcium iodate can be produced by the anodic oxidation of calcium iodide or by passing chlorine into a hot solution of lime in which iodine has been dissolved.
Calcium iodate is used as an iodine supplement in chicken feed.[1] Ethylenediamine dihydroiodide (EDDI) is a more typical source of nutritional iodine.
References
- ^ a b c d Lyday, Phyllis A.; Kaiho, Tatsuo (2015). "Iodine and Iodine Compounds". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. pp. 1–13. doi:10.1002/14356007.a14_381.pub2. ISBN 978-3527306732.
- v
- t
- e
- CaH2
- CaF2
- CaCl2
- Ca(ClO)2
- Ca(ClO3)2
- Ca(ClO4)2
- CaBr2
- Ca(BrO3)2
- CaI2
- Ca(IO3)2
- CaICl
- CaC2
- Ca(CN)2
- CaCN2
- CaCO3
- Ca(HCO3)2
- CaSi
- CaSi2
- Ca2SiO4
- Ca3(BO3)2
- CaAl2O4
- Ca3Al2O6
- Ca(MnO4)2
- CaCrO4
- CaTiO3
- CaC2O4
- Ca(HCO2)2
- Ca(CH3CO2)2
- Ca(C3H5O2)2
- CaC4H2O4
- Ca3(C6H5O7)2
- C3H7CaO6P
- Ca(C6H5O5S)2
- Ca(C6H7O6)2
- C10H11CaN4O8P
- CaC10H12O4N5PO4
- C10H16CaN2O8
- C12H22CaO14
- C14H26CaO16
- C18H32CaO19
- C36H70CaO4
- C24H40B2CaO24











